AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Western blot assay test11/23/2023 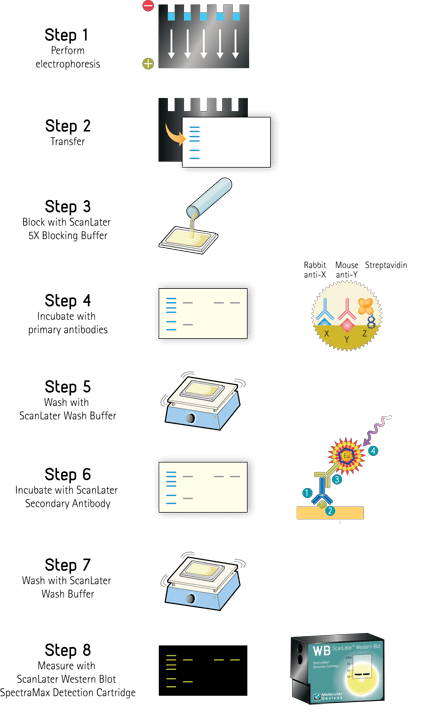
After adding the buffer, the plate is rewashed, and a selected enzyme-conjugated primary detection antibody is added. This second step is crucial because it prevents the binding of any non-specific antibodies to the plate and minimizes false-positive results. Once the incubation step is completed, the next step is to wash the plates of any potential unbound antibodies and block any unbound sites on the ELISA plate using agents like BSA, ovalbumin, aprotinin, or other animal proteins. The first binding step involves adding antigens to the plates, which are incubated for one hour at 37^oC or can be incubated at 4^oC overnight. īoth direct and indirect ELISAs begin with the coating of antigens to the ELISA plates. In a usual ELISA protocol, a serial dilution of concentrations is placed in the wells of the plate. After the results are measured, a standard curve from the serial dilutions data is plotted with a concentration on the x-axis using a log scale and absorbance on the y-axis using a linear scale. The wells are washed two or more times during each wash step, depending on the specific protocol. īetween each of the above four steps is a “wash” of the plate using a buffer, such as phosphate-buffered saline (PBS) and a non-ionic detergent, to remove unbound material. 
AP measures the yellow color of nitrophenol after room temperature incubation periods of 15 to 30 minutes and usually uses p-Nitrophenyl-phosphate (pNPP) as its substrate. The substrate for HRP is hydrogen peroxide, resulting in a blue color change. However, the substrates most commonly used are horseradish peroxidase (HRP) and alkaline phosphatase (AP). There are many substrates available for use in ELISA detection. ĭetection is carried out by adding a substrate that can generate a color. However, this method is qualitative and can determine only the presence or absence of an analyte and not its concentration. This technique can generate a color signal visible to the naked eye, with a blue color indicating positive results and red color indicating negative results. The latest development, in 2012, was an ultrasensitive enzyme-based ELISA that manipulates nanoparticles as chromogenic reporters. However, some of these techniques do not rely on using enzyme-linked substrates but non-enzymatic reporters that utilize the principle of ELISA. Further advancement in the ELISA technique led to the development of fluorogenic, quantitative PCR, and electrochemiluminescent reporters to generate signals. The first ELISA methodology involved chromogenic reporter molecules and substrates in generating observable color change that monitors the presence of antigen. Since then, the ELISA method has been used in many different applications and has become a routine laboratory research and diagnostic method worldwide. Within the same year, scientists quantified human chorionic gonadotropin in urine using horseradish peroxidase. The new method was first employed in determining the levels of IgG in rabbit serum. This was done by conjugating tagged antigens and antibodies with enzymes rather than radioactive iodine 125. The ELISA was developed by the modification of the radioimmunoassay (RIA). Two different research teams simultaneously invented the direct ELISA scientists Engvall and Perlman, and scientists Van Weemen and Schuurs. Such assays are very useful in screening, point-of-care, and home testing applications. 
In addition, enzyme conjugates coupled with substrates that produce visible products have been used to develop ELISA-type assays with results that can be interpreted visually. In addition, ELISA assays have been used extensively to detect antibodies to viruses and autoantigens in serum or whole blood. The second reagent is an enzyme-labeled antibody specific to the analyte antibody. Specific antibodies in a sample can also be quantified using an ELISA procedure in which antigen instead of antibody is bound to a solid phase. The amount of product generated is proportional to the quantity of antigen in the sample. Unbound antibody is then washed away, and enzyme substrate is added. After washing, an enzyme-labeled antibody is added and forms a “sandwich complex” of solid-phase Ab-Ag-Ab enzyme. In the most common approach to using the ELISA technique, an aliquot of sample or calibrator containing the antigen (Ag) to be quantified is added to and allowed to bind with a solid-phase antibody (Ab). This attachment facilitates the separation of bound and free-labeled reactants. In this type of assay, one of the reaction components is nonspecifically adsorbed or covalently bound to the surface of a solid phase, such as a microtiter well, a magnetic particle, or a plastic bead. Enzyme-linked immunosorbent assay (ELISA) is a heterogeneous EIA technique used in clinical analyses. Enzyme immunoassays (EIAs) use the catalytic properties of enzymes to detect and quantify immunologic reactions.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
